NEW

WINNER

NEW




NEW



Desktop power in a handheld autorefractor
Measurements in 10 seconds
Modern, intuitive operator and patient experience




Our handheld autorefractor’s combination of the open view design, wavefront aberrometry, and innovative measurement algorithms produces clinically accurate autorefraction measurements, in a durable handheld format suitable for use in clinics and in the field.
The patented PlenOptika Wavefront Refraction Engine™ precisely determines low-order refractive errors, making QuickSee Free as accurate as the high-end clinical desktop autorefractors and demonstrating excellent agreement with subjective refraction.
QuickSee Free’s technology has been clinically-evaluated in five IRB-based studies and documented in many peer-reviewed publications and conference abstracts. QuickSee Free has FDA Class I 510(k) exempt medical device registration.


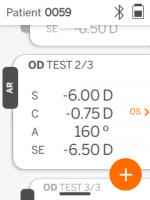

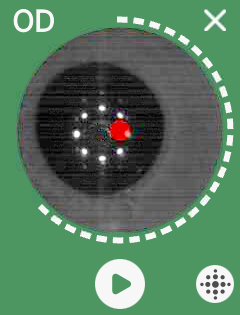
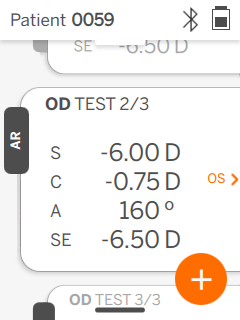
QuickSee Free’s UI is clean, intuitive, and robust


| Pupil size | 2 to 8 mm |
| Intended patient population | >= 3 years |
| Accommodation control | Open view, Fogging lens (optional) |
| Cycloplegia requirement | None |
| Dilation requirement | None |
| Illumination requirement | None |
| Acquisition time | 5, 10 seconds |
| Spherical range | -13D to +10D, increments of 0.01D, 0.125D, 0.25D |
| Cylindrical range | -8D to +8D, increments of 0.01D, 0.125D, 0.25D |
| Axial range | 0–180º, increments of 1º, 5º, 10º |
| Base technology | Wavefront aberrometry |
| Measurement modes (QuickSee Free Pro) | Simultaneous AR and K, AR only, and K only |
| Display properties | 2.4-inch LCD, capacitive touch screen, readable outdoors, true color (65,536 colors) |
| Charger properties | USB-C medical grade wall adapter, AC 100 to 240V, 50/60 Hz |
| Battery | 6 hours continuous use +/– 1 hour (10,000 mAh Li-ion); 3 hours charge time (5% - 75%); 5 hours charge time (0% - 100%); IEC 62133-2:2017 certified |
| Calibration | Factory calibrated; no field calibration needed |
| Measurement capacity | Measurement storage capacity: > 10,000 measurements |
| Regulatory classification (medical device) | Class I FDA (USA), Class IIa Product CE, MDR Compliant, Class IIa, UKCA, MDR 2002 Compliant |
| Laser safety | Class 1, IEC 60825-1:2014 certified |
| Weight | < 750 grams / 1.65 lbs |
| Dimensions | 30 cm (H) x 5.5 cm (W), 18 cm (L) |
QuickSee Free
QuickSee Free Pro Keratometry
QuickSee Free Software
QuickSee
© 2024 PlenOptika. All rights reserved. | No. RII-AEE: 14923 | No. RII-PyA: 4987
Are you ready to learn how to use QuickSee Free / Free Pro effectively? Tell us about yourself and your circumstances and we will reach out to arrange a training session with you.
QuickSee Free Pro flew on the history-making Polaris Dawn mission to measure changes in the astronauts’ vision
This website uses cookies so that we can provide you with the best user experience possible. Cookie information is stored in your browser and performs functions such as recognising you when you return to our website and helping our team to understand which sections of the website you find most interesting and useful.
Strictly Necessary Cookie should be enabled at all times so that we can save your preferences for cookie settings.
If you disable this cookie, we will not be able to save your preferences. This means that every time you visit this website you will need to enable or disable cookies again.
This website uses Google Analytics to collect anonymous information such as the number of visitors to the site, and the most popular pages.
Keeping this cookie enabled helps us to improve our website.
Please enable Strictly Necessary Cookies first so that we can save your preferences!
This website uses the following additional cookies:
Marketing performance cookies:
Website performance cookies:
Please enable Strictly Necessary Cookies first so that we can save your preferences!
More information about our Privacy Policy